We move beyond empirical observation to deep-science understanding. Our approach identifies Critical Quality Attributes (CQAs) and Critical Process Parameters (CPPs) early, ensuring every development decision is data-driven and defensible.
Establishing robust design spaces from first-in-human through commercial supply.
Pragmatic application of Quality by Design (QbD) and Design of Experiments (DoE).
Global regulatory fluency (FDA, EMA, PMDA, TGA, HC) with ICH Q8–Q10 and ISO standards integrated into early-stage strategy.
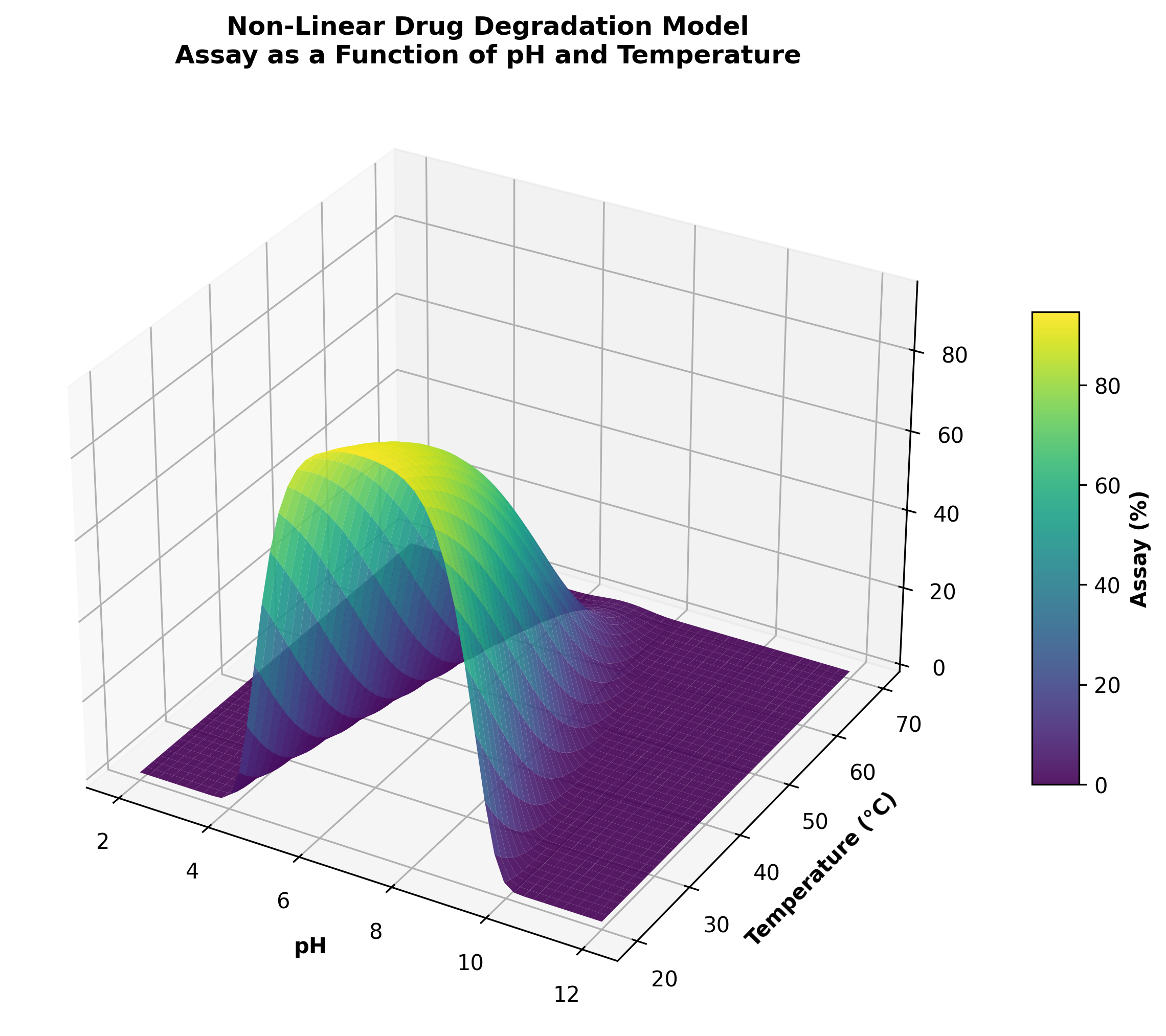
Figure 1: Multidimensional design space mapping for process robustness.
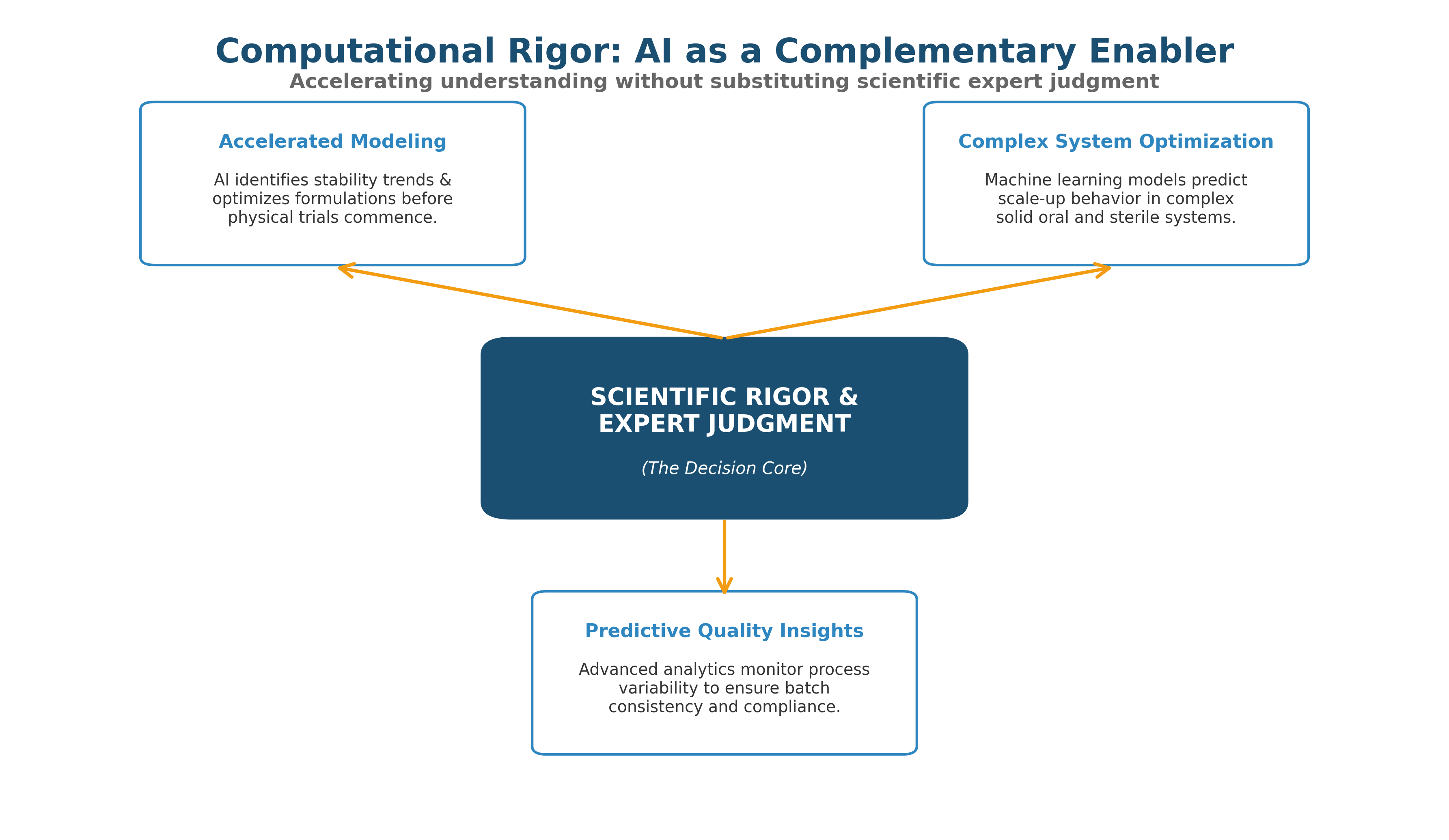
Computational Rigor, Not Substitutes
Advanced computational tools, machine learning, and AI are integrated as complementary enablers when they accelerate understanding or optimize complex systems — never as substitutes for scientific rigor and expert judgment.
Accelerated Modeling
Leveraging science and AI to identify stability trends and optimize formulations before physical trials commence.
Complex System Optimization
Predict scale-up behavior in complex solid oral and sterile systems.
Shortening Timelines While Enhancing Quality
Through in-house excellence and strategic access to best-in-class specialist partners, we solve the toughest formulation and scale-up challenges efficiently.
"Our commitment is to transform CMC drug product development into a knowledge-driven discipline that delivers high-quality medicines faster, more reliably, and more sustainably."Partner With Us