Complex Solid Oral Dosage Forms
Leveraging deep expertise in formulation and process development (BCS Class II/IV compounds), we design robust solid dose strategies that emphasize bioavailability and stability. Our approach integrates Real-Time Release Testing (RTRT) and predictive modeling to ensure commercial readiness.
- ✦ Immediate & Modified Release Dosage Forms
- ✦ Amorphous Solid Dispersions (ASD) & Lipid-Based Systems
- ✦ Statistical Formulation & Process Optimization
- ✦ Scale-Up Physics and Mechanistic Blending Models
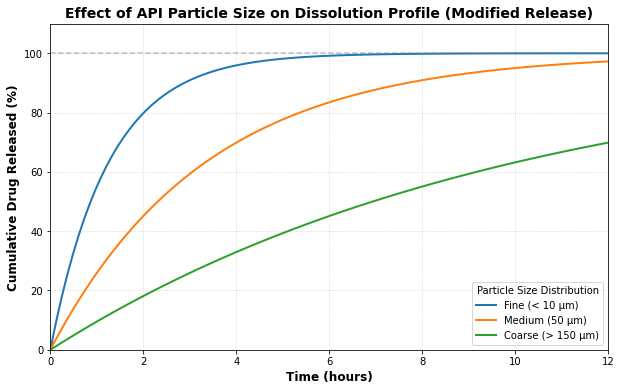
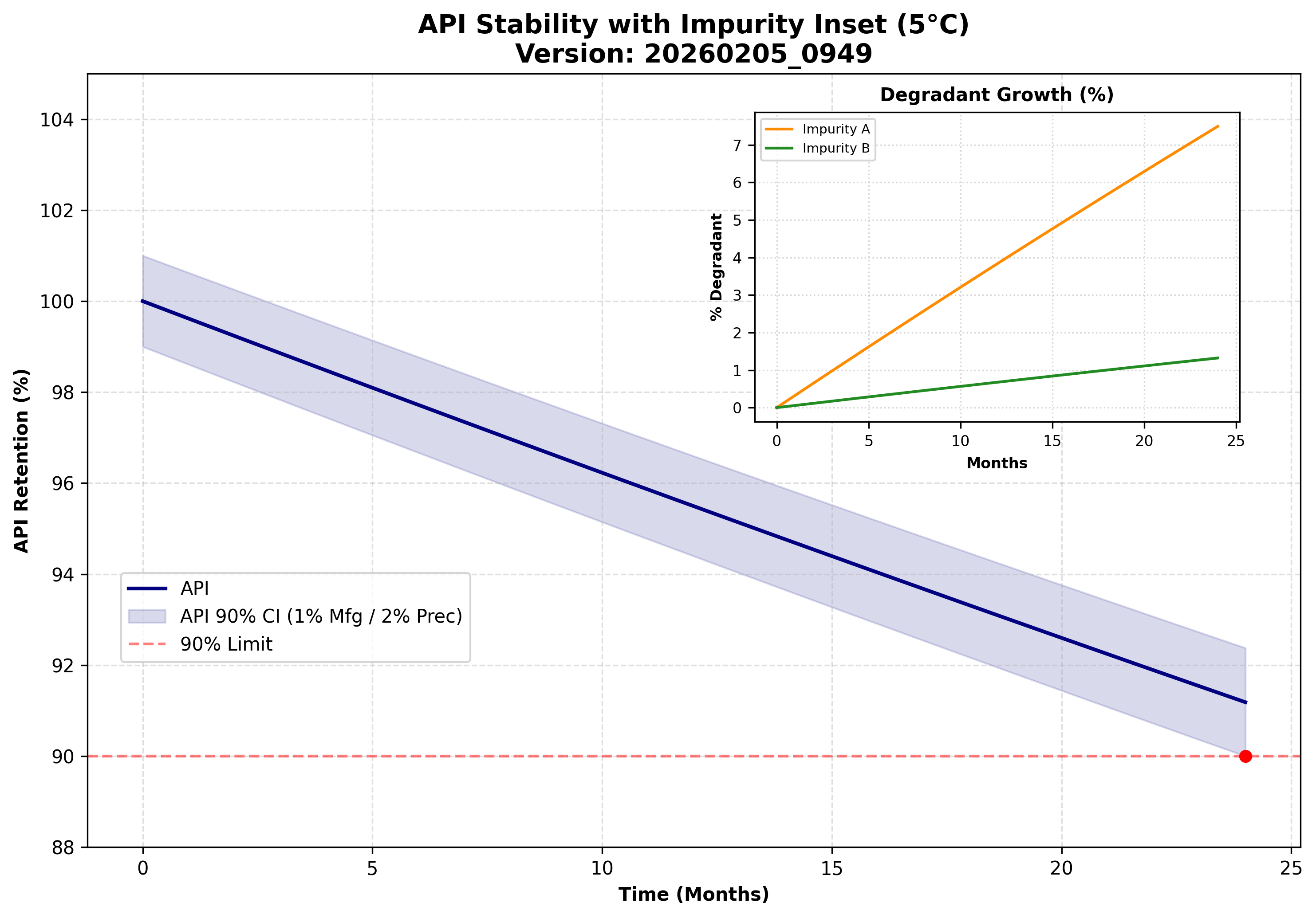
Complex Sterile Injectables
Sterile development requires a relentless focus on stability, aseptic processing, and container-closure integrity. We specialize in de-risking high-value liquid and lyophilized assets.
- ✦ Formulation of Sensitive Small Molecules and Proteins / Peptides
- ✦ Vials, Prefilled Syringes and Auto-Injectors
- ✦ Extractables & Leachables, Elemental Impurities, and Nitrosamines
- ✦ Terminal Sterilization of Thermally Sensitive Drugs
Operational Excellence in R&D & Manufacturing Sciences
Maximizing organizational agility by synchronizing resource utilization and lean workflows with integrated digital applications to accelerate the transition from molecule to market.
- ✦ Resource Utilization & Optimization
- ✦ Workflow Optimization
- ✦ Digital Strategy Development & Execution
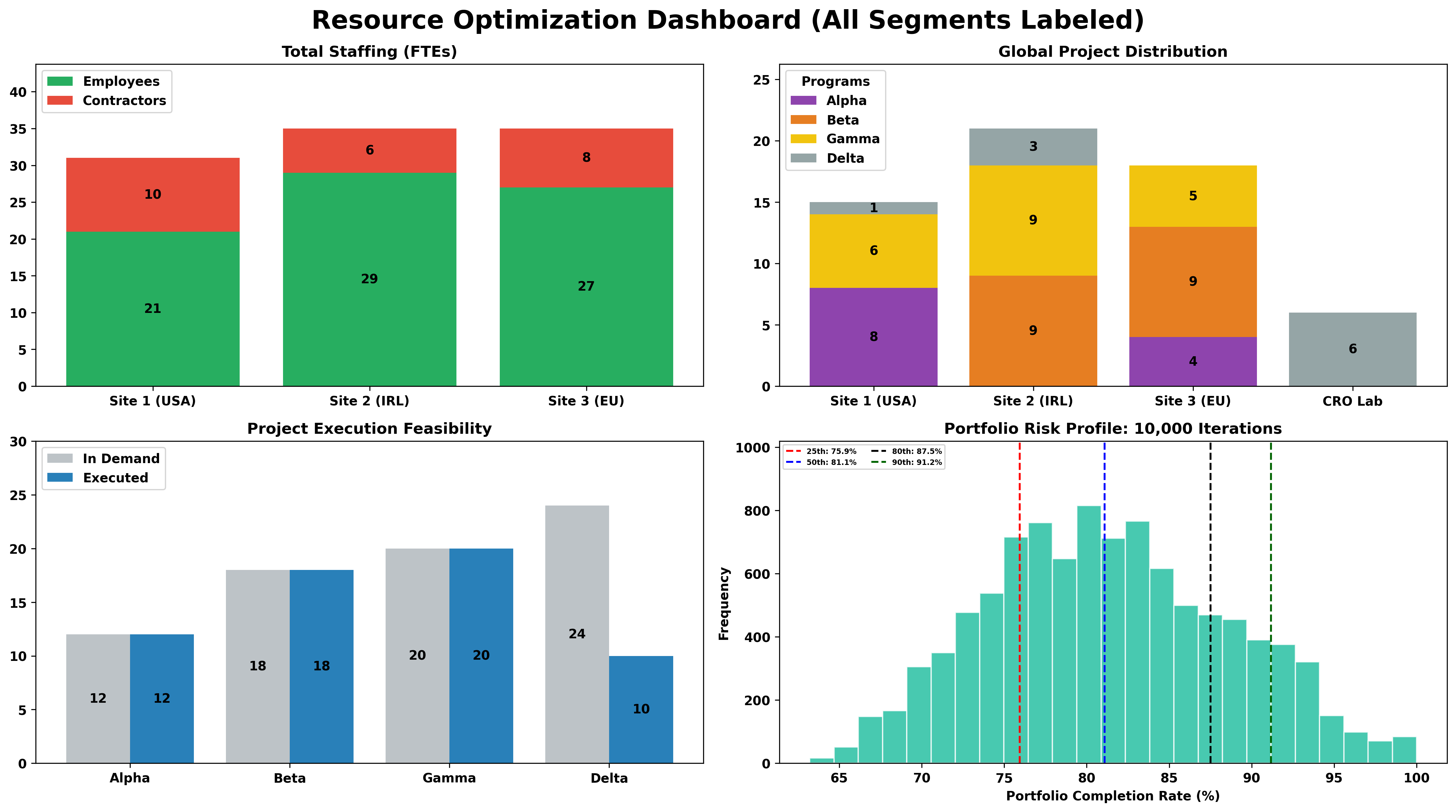
Complex exits don’t have to be chaotic
While a checklist provides the roadmap, the execution requires a steady hand to mitigate risk and protect your assets. Let’s discuss how I can manage the heavy lifting of your restructuring.
EHS • HR • Asset Recovery • GxP
Strategic Restructuring & Facility Decommissioning
"Mitigating organizational risk by orchestrating compliant facility exits and workforce transitions through rigorous EHS protocols and change management."
We provide end-to-end oversight to ensure your reputation and bottom line remain protected during periods of significant organizational change.
- ✦ Human Capital: Designing retention strategies, severance frameworks, and outplacement frameworks that honor your team and retain critical knowledge.
- ✦ Asset Recovery: Managing equipment lifecycles—from decontamination certification to high-value redeployment or resale.
- ✦ EHS Compliance: 100% compliance with hazardous waste disposal, radiation safety, and GxP documentation archival.
- ✦ Site Closeout: Coordinating utility "safe-offs" and neutralizing lab environments to meet strict lease-end obligations.