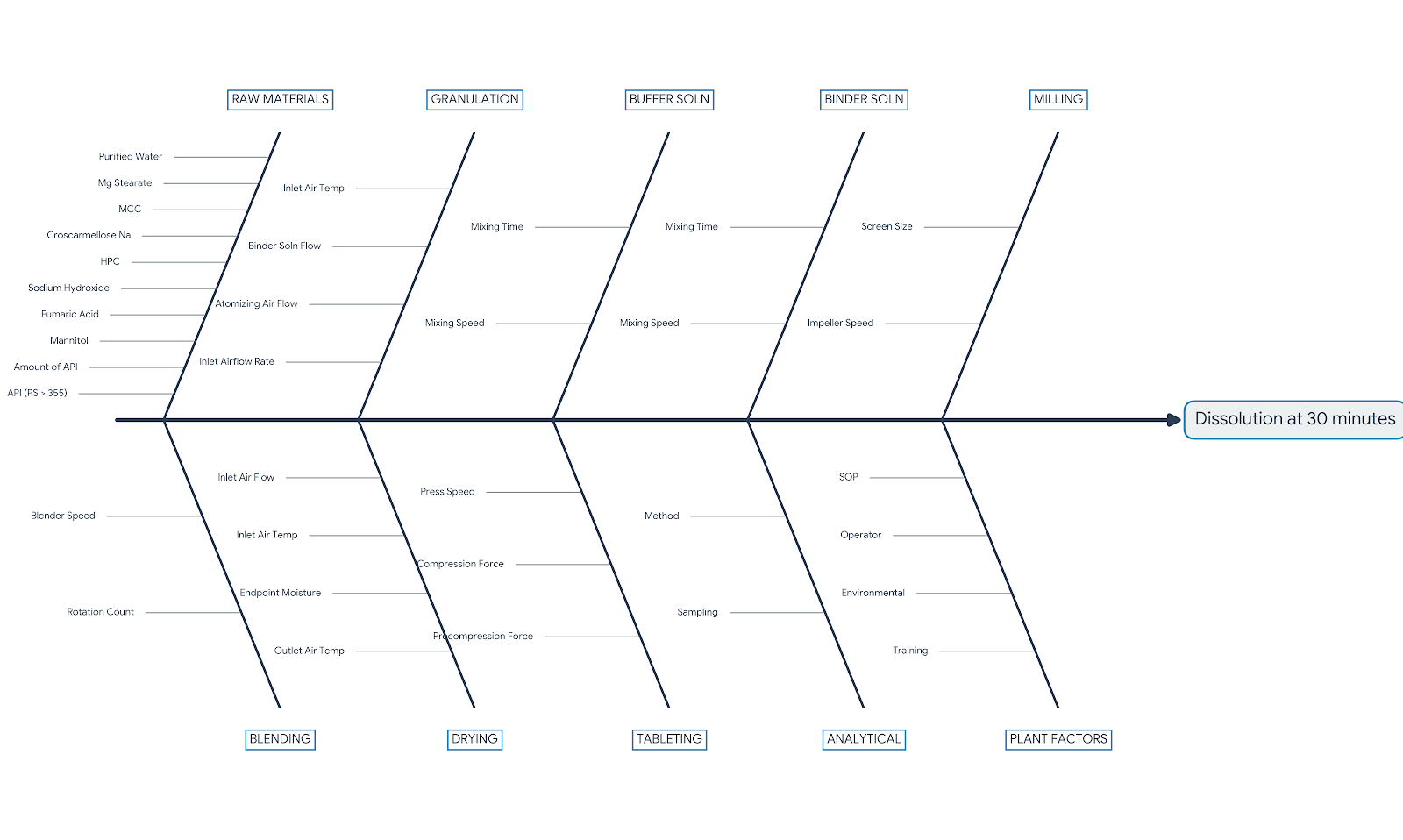
CMC Fishbone Diagram
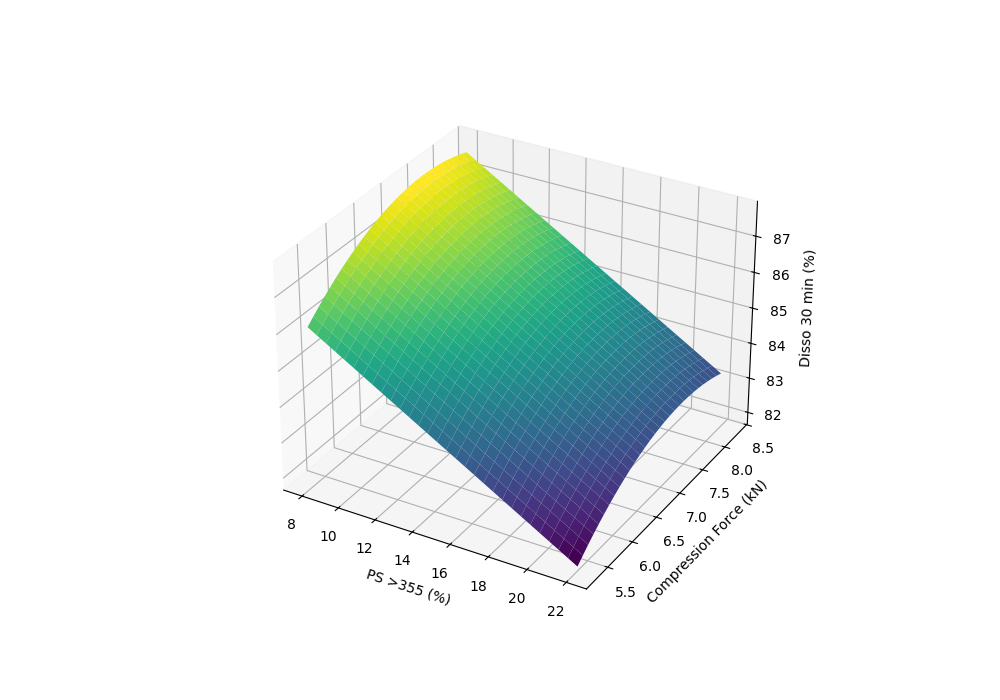
Dissolution Response Surface
Architecting a Real-Time Release (RTRT) Strategy for a Modified Release NDA
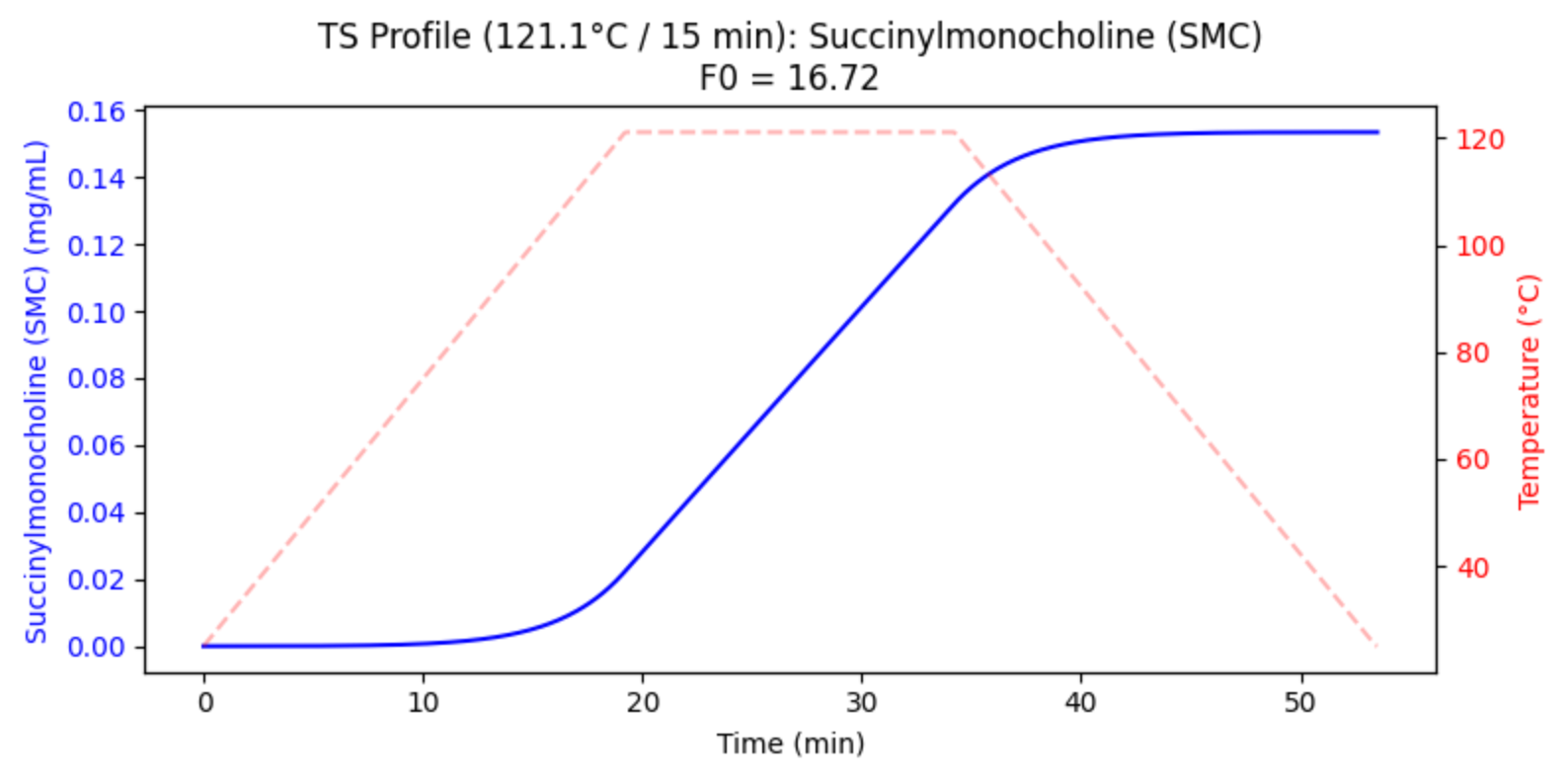
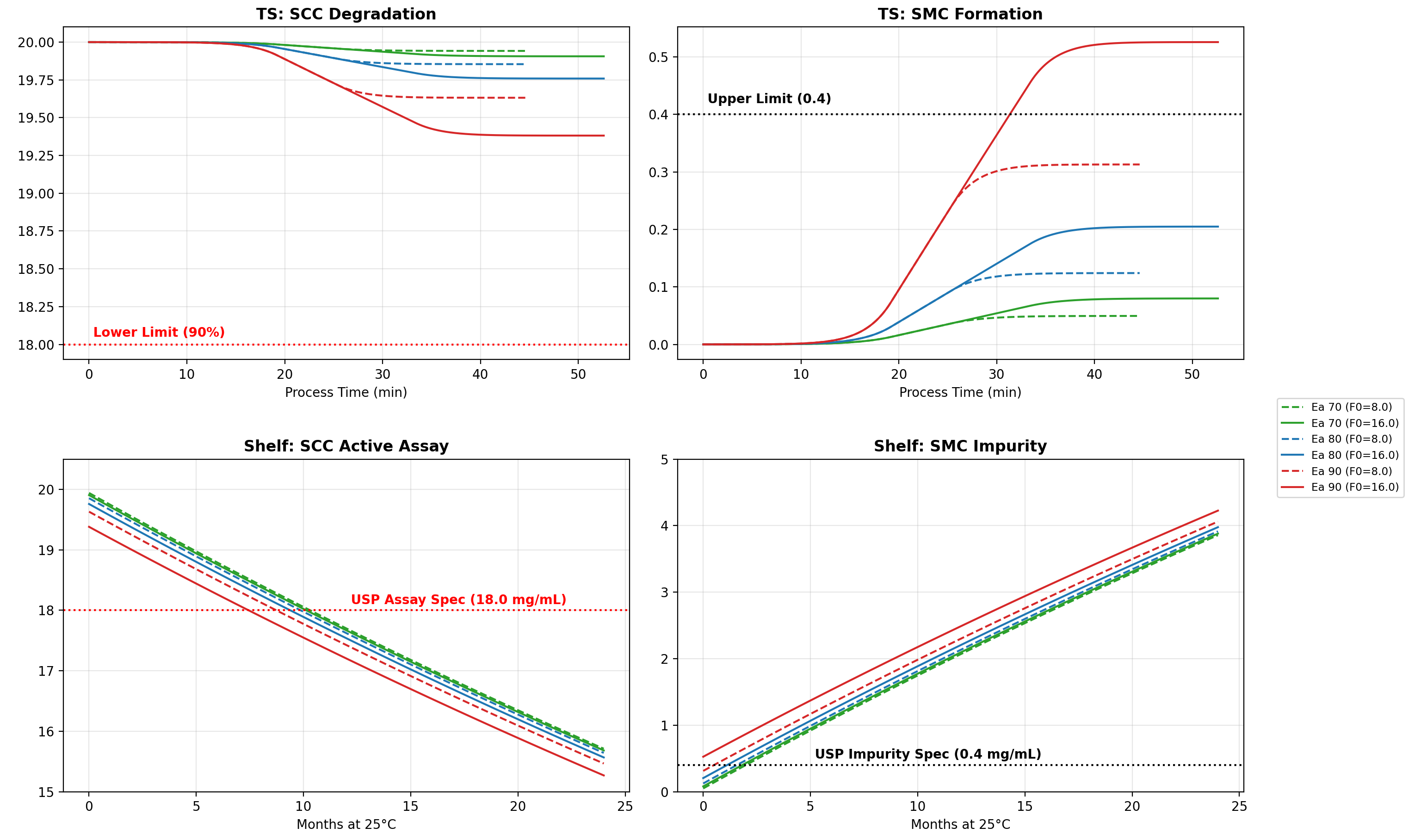
Dissolution testing is traditionally a multi-day bottleneck in pharmaceutical manufacturing. For this modified-release solid oral drug, Cachet leadership engineered a Real-Time Release (RTRT) model that predicts dissolution based on critical upstream parameters.
The Breakthrough: Equipment Independence
Through advanced Design of Experiments (DOE), we identified that Tablet Hardness served as a sufficient proxy for both compression force and press speed.